|
What’s the difference between a regular veterinary prescription and the new biosimilars? What is the cost and availability for pets?
November 12, 2018 Barbara "BJ" Andrews, SAAB Member
Breeders or pet owners, we all agree that medication costs are through the roof! Prescription drugs (human or animal) are not that costly to make but the costs of research, development and the biggie, testing and then getting FDA approval are enormous. So will these new biosimilars be any different? And what exactly are they?
According to www.Phrma.org “Biologics are medicines made from living cells through highly complex manufacturing processes and must be handled and administered under carefully monitored conditions. ... A biosimilar is exactly what its name implies — it is a biologic that is “similar” to another biologic drug already approved by the FDA.”
All non-herbal (prescription) medications are manufactured through chemical synthesis, i.e. combining specific chemical ingredients. The U.S. has been a leader in the development of ever-more prescription drugs for two reasons. Pharmaceuticals save lives and they are very profitable.
Predictions are that “the cost savings potential of biosimilars to be $54 billion over ten years”. Don’t count on that when you pull out your wallet. Development and promotional costs (such as those TV ads that bombard us) are passed on to the patient. Traditional drugs are thus very expensive and like a revolving wheel, that cost drives up prescription insurance coverage. Pet insurance is almost unattainable for dog breeders and probably won't cover any new-fangled medicines anyway.
The cost of regular prescription medications is enormous due to strict FDA regulations. That is a major reason for the ever-increasing popularity of herbs and other “natural” cures such as electrotherapy and infrared-light devices.
Biosimilars act like regular drugs but cost less to make. That’s the reason generic drugs have made headway over the last decade-because they cost less than brand-name drugs. We predict the same pricing relief from biosimilar medicines in early 2019 but don’t hold your breath waiting for a veterinary line.
In September 2018 the FDA released this statement “Congress, through the Biologics Price Competition and Innovation Act (BPCI Act) of 2009, created an abbreviated licensure pathway for biological products that are demonstrated to be biosimilar to or interchangeable with an FDA-approved biological product. FDA requires biosimilar...“ {Ref #1}
The NetPlaces Network staff did some deep digging to see if there are any biosimilar medications in the pipeline for dogs. The answer was “not really.” As of this writing only one reference popped up and it indicated that biosimilar may become important in canine cancer patients. {Ref #2} The field of biosimilars is wide open for 2019 and we can be sure that will eventually include other veterinary medicines.
Animals know what is “natural” and given opportunity they will choose the real thing over manufactured. Dogs may not know the difference between a prescription “pill” you have to hide in peanut butter or the new biosimilar medications but now YOU do!
Reference & Related Articles and Information, dig for gold below Ref #1 FDA Process In Biosimilars (offsite, copy both lines of link and paste in browser) https://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ ApprovalApplications/TherapeuticBiologicApplications/Biosimilars/default.htm
Ref #2 Passive Immunotherapy In Canine Cancer Patients (offsite, copy and paste link in browser) https://www.ncbi.nlm.nih.gov/pubmed/24755200 TheDogPress.com EST 2002 © 1811 https://www.thedogpress.com/editorials/biologics-biosimilars-for-dogs-b18A11.asp SSI
SSI Advertising Mission Statement Privacy
ii NetPlaces Network ~ Disclaimer
Brought to you by NetPlaces Network
TheDogPlace.org, world’s 1st public website, 1st online dog news, TheDogPress.com, and TheJudgesPlace.com, 1st AKC judges site
|

 First, the “what is it?”
question. A biologic is manufactured in a living system such as a microorganism,
a plant or harvested animal cells. Many biologics are produced using recombinant
DNA technology and good dog breeders know what that is.
First, the “what is it?”
question. A biologic is manufactured in a living system such as a microorganism,
a plant or harvested animal cells. Many biologics are produced using recombinant
DNA technology and good dog breeders know what that is.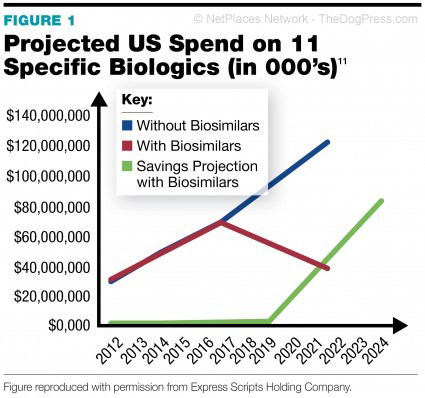 These
new medications probably won't be much cheaper due to the development and marketing
costs but they are predicted
to generate over 300 $billion in sales by 2019. Biologics are made from live
organisms and they account for 7 of the top 10 drugs today. Many of these older
drugs have or are about to come off patent and that opens the market to today’s
“biosimilars”.
These
new medications probably won't be much cheaper due to the development and marketing
costs but they are predicted
to generate over 300 $billion in sales by 2019. Biologics are made from live
organisms and they account for 7 of the top 10 drugs today. Many of these older
drugs have or are about to come off patent and that opens the market to today’s
“biosimilars”.